  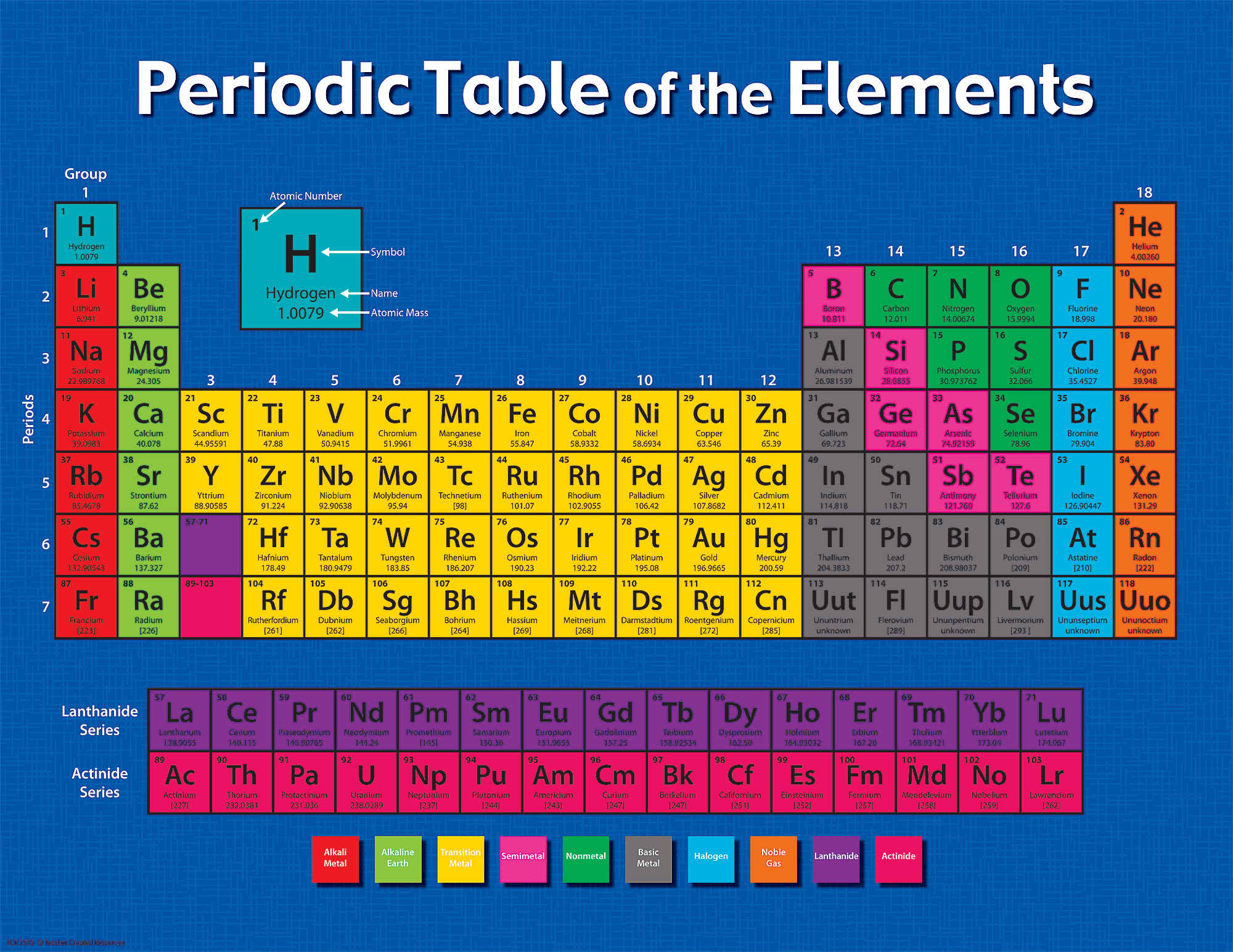 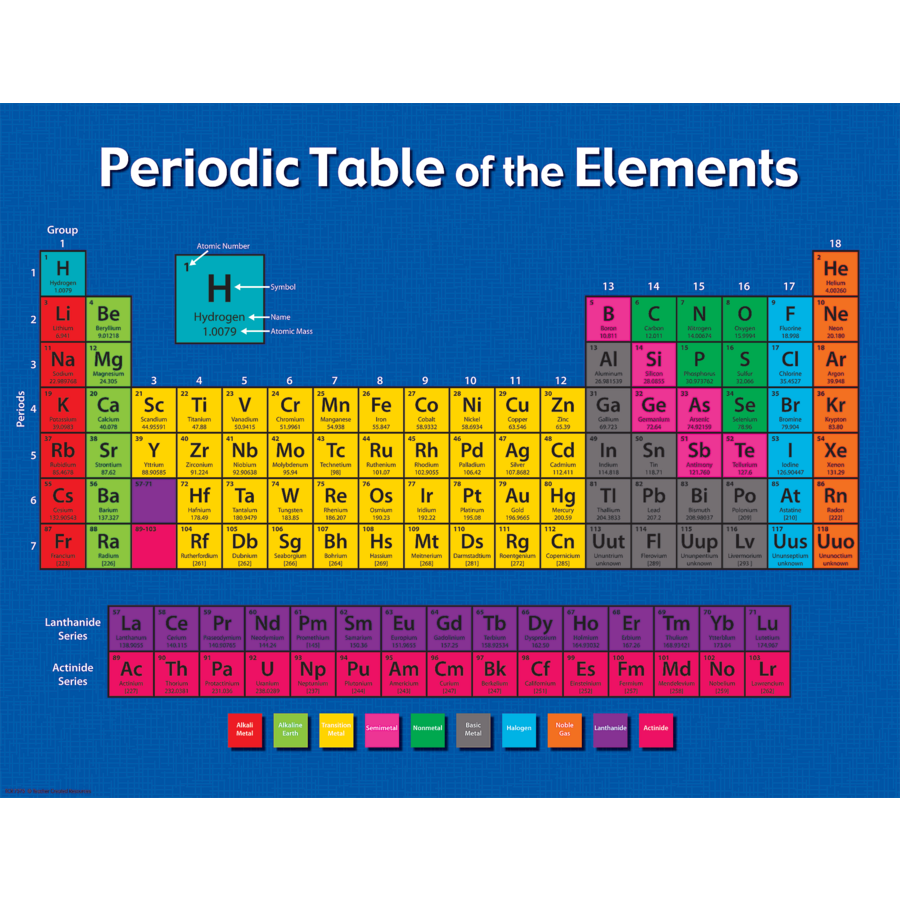
On you can find two version of the periodic table The lanthanides and actinides should actually be placed in the middle of the table as well, after lanthanum and actinium, but, because of their similarity to each other, they are removed from the middle to save space. Rows are arranged so that elements with similar properties fall into the same vertical columns ("groups"). The periodic table is a chart that organizes the elements by increasing atomic number and their chemical and physical properties (see article History of the Periodic table of elements). We can list elements in order of increasing atomic number but it wouldn't be a periodic table any more. Organization of chemical elements in alphabetical order by their names (or symbols) doesn't tell us anything about their properties but it is useful when searching for an element. Why is the periodic table shaped like it is? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
